 
At very high gas pressures (in other words, very high concentrations of gas molecules), the surface of the catalyst can be completely full of gas molecules. 
Is this unique to enzyme-controlled reactions? No! It can happen in some ordinary chemistry cases as well, usually involving a solid catalyst working with gases. If you increase the substrate concentration any more, there aren't any enzyme molecules free to help the extra substrate molecules to react. After a certain concentration of substrate is reached, every enzyme molecule present in the mixture is working as fast as it can. The reason for this is actually fairly obvious if you think about it. If you know about orders of reaction, the reaction has now become zero order with respect to the substrate. Increasing the concentration any more makes no difference to the rate of the reaction. 
In other words, at very, very low concentrations, the rate is proportional to the substrate concentration.īut as concentration increases, increasing the concentration more has less and less effect - and eventually the rate reaches a maximum. That's not our problem for this topic!įor very, very low substrate concentrations, the graph is almost a straight line - like the second chemistry rate graph above. It is easier to visualize, but involves a messy calculation to get there. Biochemists often quote it in terms of the number of molecules of substrate which a single molecule of enzyme is processing per unit time - per second, for example. In chemistry, rates are normally measured in terms of rate of change of concentration, with units like mol dm -3 s -1 (moles per cubic decimeter per second).If you have done any physics, you will know that this is a misuse of the word "velocity"! But that's what you will find in biochemistry sources, so that's what we will have to use. Biochemists talk about a reaction velocity instead of a reaction rate.Two minor things to notice before we discuss it. The graph for enzyme controlled reactions looks like this: 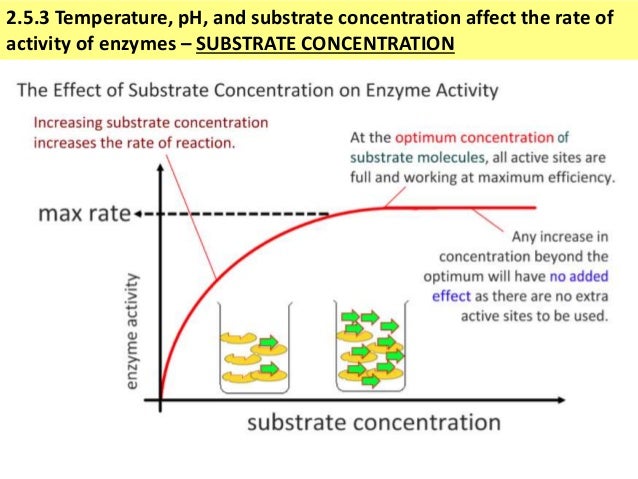
Plotting initial rates of enzyme-controlled reactions against substrate concentration 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
